|
Vegetable Oil Refining

Vegetable Oil Refining
Vegetable Oil Refining
Vegetable Oils Refining is necessary for vegetable oils & fats or animal oils & fats so they can be used for cooking and frying foods. Chemically speaking, oils are Tri-Glycerides or “Fatty-Acid Glycerin Esters” (FAGE). One Glycerin molecule combines with three Fatty-Acid molecules to form one FAGE. Oils are a blend of different types of FAGEs. The Oils can be differentiated on the basis of the length of carbon chain in the Fatty Acids and the number and location of double bonds in the carbon chains of the Fatty Acids. Greater the number of double bonds between two carbon atoms, the greater is the extent of “unsaturated” fatty-acids in the fatty-acid profile of the oils. “Unsaturated” oils and fats are considered more desirable than “saturated” oils and fats from health point of view.
What are Vegetable Oils & Fats
Vegetable Oils are obtained from a variety of seeds, grains and nuts. Most common types of oil-bearing seeds and nuts are Canola, Castor, Coconut, Corn (Maize), Cottonseed, Mustard, Olive, Palm, Palm-Kernel, Peanut (Groundnut), Rapeseed, Safflower, Soybean, and Sunflower. Other major specialty oils are obtained from Almond, Avocado, Cashew, Grapeseed, Jatropha, Pecan, Pine, Shea, and Walnut. There are literally hundreds of other seeds and nuts that are available regionally around the world.
What are Animal Oils and Fats?
Animal Oils are obtained from a variety of animals. Most common types of oils and fats include Chicken-Fat (Chicken), Fish-Oil (Fish), Lard (Pigs), and Tallow (Cows). Each one of these oils and fats comes in edible and non-edible categories and multiple grades within each category.
Why is Vegetable Oil Refined?
Vegetable and Animal Oils and Fats have impurities such as Moisture, Solids (Insolubles), Gums (Lecithins), Free-Fatty Acids (FFA), Waxes, and Compounds of Sodium, Potassium, Magnesium, Calcium, and other metals. These impurities must be removed to improve the functionality of the oils. Other characteristics of oils (such as Color, odor, and taste) are also considered impurities by modern consumers. These impurities are removed in a series of steps such as degumming (to remove gums), neutralizing (to remove FFA), bleaching (to remove color), deodorizing (to remove odor and taste), and dewaxing or winterization (to remove waxes).
What is RBD Oil?
Refined Vegetable Oil is commonly called Refined, Bleached, and Deodorized (RBD) oil. As described earlier, these are the three main steps in the refining process; although additional steps are required in most cases.
Chemical Refining
In Chemical Refining, Vegetable Oil is treated with caustic lye for separation of free fatty acids from oil. This is a conventional process that can be applied to all oils. A byproduct of alkali refining is soap-stock, which is used for manufacture of low quality washing soap. Alternately, the soap-stock can be treated with acid to recover fatty acids. The waste-water from refinery requires extensive treatment. This can be avoided by alternate Physical Refining process.
Physical Refining
In Physical Refining, Vegetable Oil is subject to distillation to remove free fatty acids. The alkali treatment is completely avoided. This reduces the amount of waste water and eliminates production of soap. This process is becoming more popular.
Degumming
The purpose of Degumming Vegetable Oils is to remove Gums. All oils have hydratable and non-hydratable gums. a. Water Degumming: Hydratable gums are removed by treating oils with water and separating the gums. The gums can be dried to produce lecithin. b. Acid Degumming: Non-Hydratable gums are removed by treating oils with acids and separating the gums.
Neutralizing
The purpose of Neutralizing Vegetable Oils is to remove Free-Fatty Acids (FFAs). Traditionally, FFAs are treated with caustic soda (NaOH). The reaction produces Soaps which are separated from the oil. Because trace amounts of soaps remain in the oil, the oil is either washed with water or treated with Silica.
Some processors prefer not to perform caustic neutralizing. Instead, they prefer Physical Refining in which the FFAs are evaporated from the oil under high temperature and vacuum. This process can be combined with deodorization step described under FFA stripping.
Physical Refining process is preferred because (a) it does not produce soaps; (b) it recovers fatty acids that provide better cost recovery; (c) there is smaller yield loss compared to caustic refining—especially for oils with higher FFAs; and (d) it is a chemical-free process.

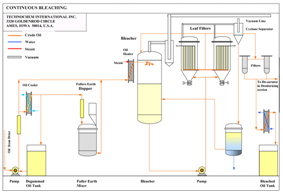
Bleaching
The purpose of Bleaching is to remove color pigments contained in Vegetable Oils. The oil is treated with Bleaching Clays that adsorb the color pigments. The clay is filtered and the clean bleached oil is stored for further processing. Process flow diagram is attached.
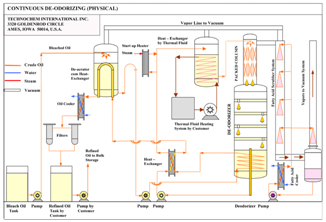
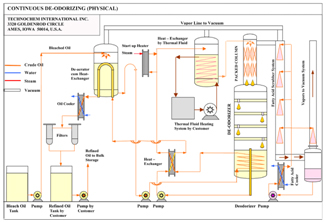
Deodorizing
The purpose of Deodorizing Vegetable Oils is to remove odor substances. The oil is subjected to steam distillation under high temperature and vacuum to evaporate all odor substances. The resulting deodorized oil is almost bland and tasteless.
FFA Stripping
As mentioned previously, the Deodorizing equipment can be modified to achieve Physical Refining. Since the conditions for deodorization and physical refining are almost identical, the modified equipment can be employed to achieve deodorization as well as physical refining.
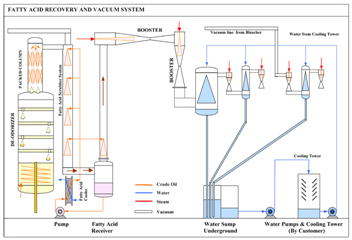
Fatty-Acid Recovery
Fatty-Acids are evaporated (distilled) in FFA Stripping. The Fatty-Acid vapors are condensed by direct contact with liquid Fatty-Acids to recover the evaporated Fatty-Acids. The distillation and condensation of Fatty-Acids is accomplished under very high vacuum to lower the boiling point of Fatty-Acids and prevent them from oxidizing.
Dewaxing/Winterizing
The purpose of Winterization Vegetable Oils is to remove waxes especially in those oils that contain waxes. Such oils are subjected to chilling and filtration to remove waxes and other high melting point substances. Winterizing is also used to separate the unsaturated fats from unsaturated fats—especially in Palm Oil or other saturated fats. The chilling process solidifies the saturated fats; thus enabling separation via filtration. 
Hydrogenation
Vegetable Oils have varying degrees of saturation levels. Some food products can only be prepared in saturated oils & fats. Hence, some food companies hydrogenate vegetable oils to produce desired level of saturation in the Oils & Fats. Vegetable oils can be hydrogenated under high temperature and pressure in the presence of a catalyst.
Bakery Fat
The bakery industry uses hydrogenated oil (i.e., shortening). However, the shortening for bakery application should be of smooth texture. Granular texture, while desired in vanaspati ghee, leads to bleeding of fat in bakery products. Hence, hydrogenated oil is cooled under controlled conditions to get a product with smooth plastic texture suitable for the bakery industry. Technochem manufactures scraped-surface heat-exchanger to produce bakery fat.
Lecithin
Gums (Lecithin) present in Vegetable Oils are valuable emulsifier and an additive in prepared foods. Hence, it may be profitable to recover them for sale to food companies. The degumming process described above produces a mixture of gums, oil, and water. The mixture of gums, oil, and water is introduced in a thin-film evaporator operating under vacuum to remove water. The resulting dry mixture of gums and oil is sold as crude dry gums or lecithin. The main design concerns are that the evaporation of water should be quick and take place under higher vacuum to prevent charring and discoloring of gums.
Soap-Splitting (Acidulation)
The caustic neutralization process described above produces Soap Stock—a mixture of Soaps (Fatty-Acid Salts), Oil, and Water. Soap Stock, by itself, is a low value by-product—usually a waste product. However, it contains considerable amount of Fatty Matter, which may be profitably recovered for sale as a higher-value product. This is accomplished by converting Soaps into Fatty Acids by reacting with an Acid. The resulting product is a mixture of Fatty Acids and Oil (commonly called Acid Oil).
Glycerolysis
The physical refining process described above produces Fatty Acid Distillate—a mixture of Fatty Acids and Oil. The value of the Distillate may be enhanced by converting Fatty Acids into Oils through Glycerolysis. This is accomplished by reacting Fatty Acids with Glycerin to produce Oil. The resulting product is dried to recover oil for sale as a higher-value product.
Oil Refining Basics
|
||||
|
||||